Introduction
Chronic endometritis (CE) and adenomyosis share many common features in pathological processes (chronic inflammatory reaction, abnormal immune response), and in terms of clinical manifestations, impacting the reproductive system and fertility (dysmenorrhea, dyspareunia, abnormal uterine bleeding, chronic pelvic pain) [1-4]. There is evidence that with endometriosis, the same as in with CE, there is a risk of implantation failure increase [5-7].
Endometriosis, including adenomyosis, is characterized by the presence of endometrial tissue outside the endometrium, with signs of inflammation in both ectopic and eutopic endometrium [8,9]. This inflammatory component worsens implantation processes, often leading to miscarriage [10-12]. Also, uterine contractility and endometrial peristalsis in uterine endometriosis negatively affect pregnancy due to the promotion of deep placentation defects, causing miscarriages and repeated pregnancy losses [13. Endometriosis can also decrease the quality of oocytes and follicular environment, affecting fertility [14].
Endometriosis has pronounced clinical signs that affect not only fertility, but also a woman's quality of life, including painful menstruation and intercourse, pelvic pain, exhaustion and discomfort due to abnormal uterine bleeding [15]. Regarding the high frequency of complaints from patients with the above-mentioned complaints, the issue of endometriosis has recently received much more attention than CE.
CE is often asymptomatic or has unclear clinical characteristics, like other inflammatory diseases of the pelvis, including endometriosis, that is detected histologically (infiltration of plasma cells in the endometrial stroma) or by immunohistochemical method (detection of the expression of syndecan-1 CD 138) during a more in-depth follow-up examination. This condition requires a complex clinical and ultrasound diagnosis, and has no specific manifestations [16,17]. CE, like endometriosis, is considered factors that induce reproductive failure. CE can also affect immune regulation and trophoblast development due to impaired endometrial receptivity. There is also evidence that the number of estrogen and progesterone receptors in the endometrium increases in women with CE [18,19].
Recently, there is a growing number of studies indicating a connection between CE and endometriosis, but the primary question still remains open and requires further study [20].
Given a number of common features between these two conditions, we decided to investigate this problem in our clinic among women who needed help in relieving their symptoms of adenomyosis (uterine endometriosis), mainly with complaints of abnormal uterine bleeding and dysmenorrhea.
During the examination, the diagnosis of adenomyosis was confirmed in a large part of the patients, and the signs of CE were also revealed. During further research, it was proposed to treat the main disease (adenomyosis), that had also CE, using traditional methods and to compare results with the results of the treatment in patients with isolated CE.
Methods
Research design
We conducted a prospective study of 108 non-pregnant patients of reproductive age at the Kyiv Maternity Hospital № 1 (Ukraine), the clinical base of the Obstetrics, Gynecology and Perinatology Department of the P.L. Shupyk National Healthcare University of Ukraine who presented with abnormal uterine bleedings and dysmenorrhea in 2020-2021. Organic pathology was excluded, taking into account the recommendations and classification of FIGO PALM-COEIN [21]. All patients were examined, and an aspiration biopsy of the endometrium was performed, to prescribe further recommendations. The obtained endometrial tissue samples were fixed in 10% buffered formalin and transported to the pathomorphological unit for further histological and immunohistochemical examination.
The patients were diagnosed “adenomyosis” considering the MUSA criteria after clinical and sonographic examinations. Most patients showed signs of diffuse adenomyosis. The diagnosis of adenomyosis was confirmed through histological analysis of biopsy samples. CE was confirmed by histological and immunohistochemical methods.
All studied women were of reproductive age, did not use oral contraceptives, progestins and antibiotics during the last 6 months. Among the studied women, neoplastic lesions and sexually transmitted infections were excluded (using the polymerase chain reaction (PCR) method).
Descriptive histology
During the histological examination of microsections stained with hematoxylin and eosin, CE was confirmed by the presence of stromal infiltrate, with a predominance of lymphocytes and plasma cells with "spindle-shaped stromal cells", and focal moderate inflammatory infiltration was revealed.
The following criteria was used to diagnosis of adenomyosis [22]:
- Presence of endometrial glands and stroma at a distance >2.5 mm from the basal layer of the endometrium;
- Presence of proliferative changes and an absence of secretory changes in ectopic glands;
- The reaction of the myometrium in the form of hyperplasia and hypertrophy of muscle fibers in the area of endometrioid ectopy.
Immunohistochemical diagnosis
The gold standard for the diagnosis of CE is an endometrial biopsy, which reveals infiltration of plasma cells in the endometrial stroma due to syndecan-1 CD 138 expression. The threshold value for CD138 (a plasma cell marker) in the immunohistochemical diagnosis of CE was ≥1 CD138 positive cells per 10 high-power fields (at 400x magnification).
Statistical analysis
Statistical analysis was performed using the statistical package Stata 12. The distribution of individual clinical parameters in groups with/without the presence of adenomyosis was compared using Fisher's exact test. When appropriate, the Mann-Whitney U test was used to assess the probability of a difference in quantitative parameters between groups with a preliminary assessment of the normality of the distribution of the relevant characteristics in the studied groups. A p value of < 0.05 was considered as significant.
Results
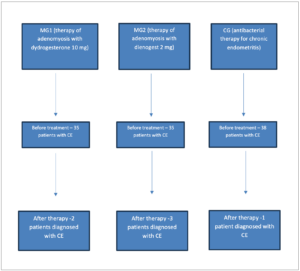
A total of 108 non-pregnant women with CE participated in the study, among them 70 patients were diagnosed with uterine adenomyosis. They were divided into three groups: main group 1 (MG1), main group 2 (MG2) and control group (CG). MG1 included 35 women with adenomyosis and CE who subsequently received adenomyosis therapy with dydrogesterone 10 mg twice per day from the 5th to 25th day of the menstrual cycle for 3 months. MG2 included 35 women with adenomyosis and CE who received adenomyosis treatment with dienogest 2 mg daily for 3 months. All other patients with CE (n=38) who received empiric antibacterial therapy with doxycycline 200 mg daily for 14 days were included in the CG. Such a division of groups was carried out to evaluate the effectiveness and feasibility of adenomyosis treatment in the case of combination with CE, as well as to compare hormonal therapeutic approaches in women with adenomyosis and CE with antibacterial therapy of CE.
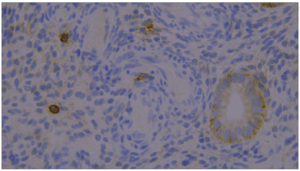
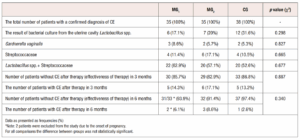
Figure 1 (Figure 1) shows the image of the immunohistochemical study from the material of a patient with CE of the CG. The endometrial sample was obtained by Pipelle biopsy. The main data characterizing the studied groups, the results of bacteriological culture from the uterine cavity, as well as the evaluation of the results 3 and 6 months after treatment are presented in Table 1 (Table 1). As one can observe, patients of all studied groups were CE positive. The number of participants at the end of the study changed because 2 patients of the main group 1 (MG1) (those treated with dydrogesterone) became pregnant within 6 months of therapy and were excluded for the comparison analyses.
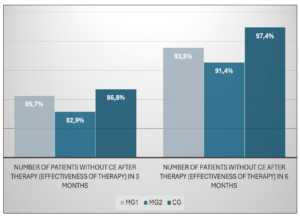
Diagram showing the distribution of patients grouped according to the type of therapy and the results are shown in Figure 2 (Figure 2). After 3 months of treatment, 85.7% (30 women) in Group 1 and 82.9% (29 patients) in Group 2 were negative for CE, compared to 86.8 % (33 patients) in the CG. The difference between all groups was not statistically significant (p=0.887). After 6 months of treatment, 93.9% in MG1 (31 women out of 33, the total number of subjects changed due to the exclusion of 2 patients from the study), 91.4% in MG2 (32 women) and 97.4% in CG (37 patients) were negative for CE. The difference between groups was also not significant (p=0.340) (Figure 3).
Discussion
The present study found that among reproductive-aged patients with CE (without sexually transmitted infections) combined with adenomyosis, the effectiveness of hormonal therapy of adenomyosis was similar to that of antibacterial therapy for CE alone.
Given the severity of the symptoms that disturb a woman with adenomyosis and the impact on her general and psycho-emotional state, as well as social activity (painful and abundant menstruation, anemia due to bleeding, chronic pelvic pain, negative impact on fertility), timely diagnosis and treatment of this disease is extremely necessary.
Our results suggest that in cases in which adenomyosis and CE are both present, the administration of dydrogesterone or dienogest is equally effective, with no need for antibacterial therapy.
Currently, a number of studies have been conducted that show the connection of adenomyosis with reduced fertility [23,24]; and the treatment of this condition is associated with an increase in pregnancy and birth rates [25]. There are also studies analyzing the effect of dydrogesterone therapy for endometriosis, which show that the use of this drug reduces pelvic pain, dysmenorrhea, increasing the frequency of pregnancy and reducing the risk of some side effects [26].
In our study, 2 pregnancies out of 35 women (5.7%) occurred within 6 months after completion of adenomyosis therapy with dydrogesterone, data of these patients were excluded from analysis. This may also indicate an additional positive effect on fertility in women who have adenomyosis and CE and requires further research.
Funding
None.