Introduction
Congenital uterine anomalies (CUA) can be a cause of infertility in females. However, the majority of these anomalies go unnoticed after either puberty or when a woman of childbearing age has difficulty conceiving due to repeated adverse pregnancy outcomes. In fact, 3 to 5% of CUA are diagnosed over the age of 16, with the majority of them presenting with amenorrhea. The pathogenesis of these developmental anomalies arises due to disturbances of the Müllerian duct development during fetal growth [1,2]. Specifically, at embryonic development, two sets of paired ducts arise at around 6 weeks post fertilization, the mesonephric (Wolffian) and paramesonephric (Müllerian) ducts. At this time, the primitive gonads are indistinguishable by sex and as development occurs from weeks 8 to 12, the paired Müllerian ducts separate to form the fallopian tubes, the cervix, the uterus, and the superior portion of the vagina [3]. Due to inadequate reporting, an accurate assessment of the prevalence of CUA may be difficult. However, reported prevalence is known to differ based on patient population. A previous systemic review demonstrated that CUA was 5.5% in an unselected population, 8% in infertile patients, 12.3% in patients with a history of miscarriage, and 24.5% in patients with miscarriage and infertility [4]. Mullerian malformations are anomalies that arise due to errors in the prenatal and postnatal development of the paramesonephric duct.
Many major CUAs are a risk factor for poor reproductive outcome [5]. It is therefore, imperative, attributable to psychological and/or physical consequences, to assess the presence of congenital anomalies correctly, particularly in females diagnosed with infertility and/or recurrent pregnancy losses [6,7]. We present a case of a young female patient found to have a hypoplastic uterus upon evaluation for chronic pelvic pain.
Case presentation
An 18-year-old G0 female presented to the vulvovaginal clinic for evaluation of chronic pelvic pain. She had a history of amenorrhea and pelvic pain since the age of 16, which was unresponsive to NSAIDs. Her past medical history showed an eating disorder, and reports eating only once a day with perfectionistic habits. Physical exam findings revealed a low body mass index (BMI: 15 k/m2), small breasts, normal pubic hair development, a hypoplastic vagina, and small cervix.
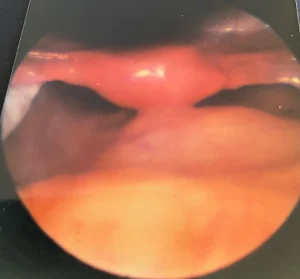
Her workup for chronic pelvic pain resulted in a negative pregnancy test; karyotype 46 XX; complete blood count and metabolic panel: mild anemia and elevated liver enzymes; low FSH, LH, and estradiol; normal TSH, free and total testosterone and prolactin; and low bone mineral density. Evaluation of chronic pelvic pain included a pelvic ultrasound, which revealed a hypoplastic uterus. The ovaries were also hypoplastic; the endometrium was not assessed. A diagnostic laparoscopy was performed with a differential diagnosis of endometriosis. Neither were found, however a hypoplastic uterus was found with the characteristic appearance shown in Figure 1, confirming the diagnosis of Mayer-Rokitansky-Küster-Hauser Syndrome (MRKHS).
The patient was therefore referred to Psychiatry for the confirmation and management of her eating disorder. Additionally, she was referred to Reproductive Endocrinology to evaluate and treat her primary amenorrhea and was counseled about her condition and how it affects her sexual health. Finally, she was prescribed Amitriptyline 25 QHS, Gabapentin 10 mg AM and Effexor 75 extended-release AM for her chronic pain. Despite this, none of the suggested referrals was completed since she moved to another country; consequently, no follow-up is available.
Discussion
There is no consensus on the classification of CUAs; however, one of the most used classifications is that which was adopted by the American Society of Reproductive Medicine [4]. Class 1 of this classification corresponds to the agenesis or hypoplasia of the uterus and the vagina. Its extreme form is known as the MRKHS. It presents with agenesis or severe uterine hypoplasia and the absence of the upper 2/3 of the vagina in patients with normal female karyotype (46, XX), normal female secondary characters, normal ovarian functions and underdeveloped vagina [8]. These features are for the most part in agreement with the findings of our case. MRKHS is the second most common cause of primary amenorrhea after gonadal dysgenesis. The familial distribution of the cases of MRKHS suggests a genetic link in etiology. Multiple genes contribute to the normal development of the female reproductive system, including the HOXA and WNT4 genes. A mutation in either of the developmental genes that regulates growth and differentiation during embryogenesis can cause abnormal development and cause uterine anomalies [9]. MRKHS is subdivided into typical or type A which has symmetric muscular buds, normal fallopian tubes and atypical or type B which manifests with asymmetric muscular buds, abnormal fallopian tubes, extra gynecological issues such as renal, skeletal, hearing, cardiac, and ocular anomalies [8]. Our case obviously belongs to type A as none of these mentioned extra gynecological findings was evident.
The findings in our case, of an 18-year-old GO female that presented chronic pelvic pain, were: a hypoplastic uterus, ovaries, vagina, and a small cervix and a karyotype of 46, XX. karyotype; low FSH, LH, and estradiol; normal TSH, free and total testosterone and prolactin which are consistent with the diagnosis of MRKHS. There was no evidence of endometriosis. Also, the absence of increased height compared to female counterparts and any evidence of intra-abdominal testicles ruled out the different diagnosis of the androgen insensitivity syndrome. However, in other reports, low levels of FSH, LH and estradiol are inconsistent with the findings of MRKHS [8]. The explanation we propose for the finding of low FSH, LH and estradiol in our case is due to the eating disorder that obviously resulted in a low BMI of 15. Studies have shown that in patients with anorexia nervosa there is an alteration in the 24-hour secretion of gonadotropins with decreased pulses of FSH and LH [10].
In terms of prognosis and treatment, patients with a hypoplastic uterus are initially diagnosed most commonly through magnetic resonance imaging or an ultrasound. Although infertility is a commonly cited issue among patients with this condition, certain treatment options may exist such as hysteroscopic metroplasty mostly in patients with pregnancy loss and reproductive difficulties [11]. Postoperative hysterosalpingograms have confirmed the efficacy of the treatment in many cases [12].
When addressing the psychosocial stressors and quality of life present in patients diagnosed with MRKHS, an array of mental health challenges can accompany the individual. With a high rate of infertility, patients often feel anxiety and loss of confidence [13]. Patients may also feel that they are unable to fulfill the conventional “female sex role” as defined by society, further leading to potential anxiety and depression [13]. Proper education and counseling are key with patients early on in the diagnosis to aid in their understanding of the condition and potential treatment options, such as creating a neovagina.
Even though a definitive diagnosis of anorexia nervosa has not been made in this patient, her history of restrictive food intake and low BMI raises concern for this diagnosis. This is important in this case since ultrasonographic studies in post pubertal women with anorexia nervosa have demonstrated that after significant weight loss, the uterus regresses back to its prepubertal length and the endometrial stripe appears very thin or even undetectable [14]. The same study also demonstrated that in such instances, the ovaries also regress to their prepubertal, immature size and may not be visualized. Did the eating disorder found in this patient worsen her case of hypoplasia of the uterus, ovaries, and the vagina or was the psychological stress associated with CUA contributory to her eating disorder? The answer to these questions can be established with more detailed research. As earlier stated in this report, low self-esteem is one of the findings in patients with CUA. The patient in this case seemed to have had low self-esteem as could be surmised from her eating disorder. Researchers have established a strong association between eating disorder symptoms and certain aspects of self-image [15]. In our case, we hypothesize that the chronic pelvic pain observed may be related to functional gastrointestinal disorder (FGID). This is consistent with the findings of Abraham & Kellow who established that the gastrointestinal symptoms common in patients with eating disorder demonstrate the presence of FGID [16]. They also concluded that factor analysis of gastrointestinal symptoms in patients with eating disorders are generally consistent with the Rome II classification of FGIDs. Although our patient presented with chronic pelvic pain, the absence of painful menstruation, dyspareunia, or malformation of the upper urinary tract effectively ruled out unicornate uterus [17].
Conclusion
We presented the case of an 18-year-old GO female who initially presented with chronic pelvic pain, which after an extensive work up was diagnosed with MRKHS. The patient also had an eating disorder, which we believe has led to low levels of gonadotropins contrary to the typical finding in patients with MRKHS.
Funding sources
The authors have no funding sources to declare.
Acknowledgments
We would like to appreciate the help of Evangelyna Nguyen with editing and reviewing the case report. We are also grateful to the Texas Tech University Health Sciences Center at the Permian Basin, Texas, USA.
Authors contributions
Ventolini GV consented the patient and drafted the case report and contributed to the conception, design, and writing of the case report. AS, RE, KG, and JG contributed in the writing and editing of this case report. All authors have read and approved the final version of this case report.
Authors disclosure of potential conflict of interest: All authors declared that they have no conflicts of interest.
Patient consent: The patient consent form was obtained.