INTRODUCTION
The association between maternal vitamin D levels and fetal growth is a matter of debate. A systematic review concluded that evidence in favor of a direct relationship is conflicting because of the heterogeneity of existing studies [1]. A series of physiological mechanisms, though, supports the association. The promoters of some amino acid transporter genes include vitamin D response elements, and maternal levels of vitamin D are positively associated with the expression of genes implicated in amino acid transport in the placenta [2]. Whether supplementation with vitamin D may have an effect, however, has been poorly investigated. A study in India, where hypovitaminosis D is highly prevalent, found that vitamin D supplementation improved weight, length and head circumference in the newborn [3]. However, a subsequent randomized controlled trial, the Maternal gestational vitamin D supplementation and offspring bone health (MAVIDOS) study, did not detect any effect on whole-body bone mineral content and somatometric features at birth [4].The growth and mineralization of the fetal skeleton requires adequate transplacental transfer of calcium. This process is favored by increased intestinal calcium absorption in the mother, an important function of vitamin D [5]. However, the effect of associating calcium with vitamin D in pregnancy has been poorly investigated, with outcomes restricted to the impact on preeclampsia, gestational diabetes, preterm birth and neonatal death [1]. There are, however, some indirect indications that calcium supplementation might have an effect on birthweight [6].Thanks to the possibility of exploring, through high-resolution three-dimensional ultrasound, how growth evolves along fetal life, it has been confirmed that femoral growth from 19 to 34 weeks of pregnancy is a critical period that strongly associates with proximal femoral geometry at 6 years. This suggests a long-term predictive potential in relation to later skeletal growth [7]. Studies using that technology have shown that the mother’s circulating levels of vitamin D are associated with the distal metaphyseal cross-sectional area [8], and with femoral volume at 34 weeks of pregnancy [9]. We designed a randomized study to investigate whether supplementation with vitamin D3 (cholecalciferol) plus calcium along that specific pregnancy window would have an impact on femoral growth and geometry, as measured with 3D ultrasound at mid-gestation, and on birthweight.
METHODS
Patients and explorations
Women at the 12th week of pregnancy attending their first prenatal screening visit at our center, which serves a population of low socioeconomic level, were invited to participate. The inclusion criteria were age 18-45 years, no accompanying comorbidities, and a body mass index (BMI) between 20 and 40 kg/m2. Women whose pregnancies were obtained through assisted reproductive technology or with multiple pregnancies were not eligible.
Upon acceptance, a structured anamnesis and a basic exploration were performed. A first routine ultrasound assessment was carried out with the purpose of confirming the gestational age and completing the prenatal study. Women were instructed to continue using the poly-vitamin preparation prescribed at their primary care center, which included 200 UI cholecalciferol daily, and were given an appointment for a further visit at the 20th week. All participants were using the same preparation, as per protocol in the health department to which our center refers.
At the further visit, an analytical study was performed in addition to the routine clinical checks and basic exploration. Blood for analysis was drawn between 08.00 and 10.00 a.m. after an overnight fast. Basic biochemical parameters were analyzed using enzymatic methods with an auto-analyzer (Olympus AV 5200; Tokyo, Japan). Vitamin D was quantified with the Elecsys vitamin D total assay (Roche Diagnostics International, Totkreuz, CH), which measures 25-hydroxyvitamin D (25OHD) by an electro-chemiluminescence binding procedure. The Elecsys coefficients of variation were £6.5% (within-run) and £11.5% (between-run). Three sub-categories were established according to the circulating levels of 25OHD: deficient when <10 ng/mL, insufficient when 10-20 ng/mL, and sufficient when >20 ng/mL [10]. At this same appointment, women were assigned, according to a randomized, unblinded protocol, to either usual care (UC group) or to the calcium plus vitamin D (CD group) treatment. Treatment in the CD group consisted of calcium carbonate, in a dose equivalent to 1200 mg elemental calcium, plus 1000 IU cholecalciferol divided into two doses, at breakfast and dinner, each day. Women were seen again at the 30th week. An ultrasound assessment of fetal femoral volume was performed at both the 20th-week and the 30th-week visits. Adherence to the treatment was ascertained from the women’s self-reports, which were verified against prescription reports recorded by their primary care physicians, visible in their electronic charts.
The study was approved by the ethics committee at our center. Informed written consent was obtained from each participant, who had first been informed of the purpose of the study.
Ultrasound measurement of fetal femoral volume
An experienced operator explored the fetal femur with a Voluson 730 Expert (GE Healthcare, Chicago, IL, US) machine fitted with a curvilinear 3D transducer, with a frequency range of 2 to 5 Mhz. The multiplanar imaging mode was used to identify anatomical structures and optimize the anatomical slices. The splaying index, as described by Mahon et al. [8], was defined as the ratio between an anatomical landmark consisting of the distal cross-sectional metaphyseal area (DMA), expressed in cm2, and the femoral length (FL), expressed in cm.
Both the FL and the DMA were measured in triplicate and the corresponding mean results were taken as the values for analyses. The 20th and the 30th week replicate assessments in 25 pregnancies were used to calculate precision. In our hands, the coefficients of variation for FL were 0.7% at 20 weeks and 0.6% at 30 weeks, whereas those for DMA were 4.2% and 3.8%, respectively.
Statistical analysis
Continuous data were analyzed by applying Student’s t-test, analysis of variance (ANOVA), and analysis of covariance (ANCOVA). Proportions were analyzed by chi-squared test after applying Yates’ continuity correction in 2x2 tables. The covariates age, BMI and parity were included in ANCOVA tests in order to control for the potential effects of these confounders. The Kolmogorov-Smirnov one-sample test was used to check whether variables were normally distributed. If the normality assumption was violated, logarithmic or square root transformation of the variable was applied to induce normality. The Levene test was applied to assess the null hypothesis that variances were homogeneous between groups. Statistical significance was defined as p≤ 0.05. Unless otherwise specified, the values shown in the text, tables and figures are means ± SEM. The entire statistical analysis was performed using the Statistical Package for the Social Sciences (IBM SPSS Statistics, version 22).
RESULTS
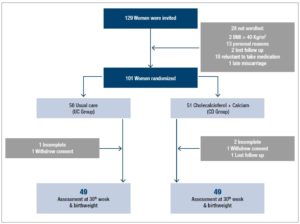
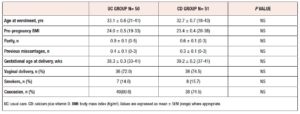
A total of 98 out of the 101 initially randomized women completed the trial. Figure 1 shows the flowchart of the study, in which 49 women completed each of the protocols. The distribution of the delivered infants [males (n=46) and females (n=52)] was even between the groups. Table 1 shows that the baseline clinical characteristics of the women were similar between the groups. The ethnic origin of the women was mainly Caucasian (n=78, 77.2%), the remaining 23 being Latin American mestizo (n=18) and Asian (n=5) women.
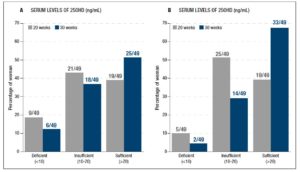
The baseline levels of 25OHD were similar between the UC group (18.7 ± 1.3, range 4-45, ng/mL) and the CD group (18.6 ± 1.2, range 3-36 ng/mL). The intervention led to an increase in the CD group, which reached 24.9 ± 1.3 (range 7.5-51.5) ng/mL vs the 20.9 ± 1.4 (range 3.1-41) recorded in the UC group (p= 0.049). The ratio between the 30th week and the 20th week values was also significantly different between groups (p= 0.004). Figure 2 shows how the three categories (deficient, insufficient and normal) were distributed in each group at the 20th week and at the 30th week. The increase was not accompanied by changes in the serum levels of total calcium and phosphate (data not shown).
The FL at 20 weeks was similar between the groups, being 3.62 ± 0.03 cm in the UC group and 3.57 ± 0.04 cm in the CD group. Similarly, there were no significant differences in the corresponding values at the 30th week: 5.22 ± 0.04 cm vs 5.18 ± 0.04 cm, or in the 30th week: 20th week ratios, which were 1.448 ± 0.017 (UC group) and 1.455 ± 0.015 (CD group).
The DMA at 20 weeks was 0.26 ± 0.10 cm2 in the UC group and 0.29 ± 0.01 cm2 in the CD group. The values increased in both groups, to reach 0.55 ± 0.02 cm2 in the UC group and 0.62 ± 0.01 cm2 in the CD group at 30 weeks. The ratio between the values recorded at the 30th week and the 20th week, which proved more informative of the progression in both groups, was 2.22 ± 0.09 in the UC group and 2.20 ± 0.09 in the CD group, a non-significant difference.
The splaying index (cm/cm2), which integrated FL and DMA, was 0.07 ± 0.01 and 0.11 ± 0.01 in the UC group, and 0.08 ± 0.01 and 0.12 ± 0.01 in the CD group at the 20th and the 30th weeks, respectively. As for the DMA, two groups showed similar ratios between the splaying index values recorded at the 30th week and the 20th week.
Delivery occurred at a similar gestational age in both groups. Birthweight did not differ significantly between the groups, being (mean ± SEM, g) 3,365.9 60.3 (range 2,220-4,120) in the UC group and 3,350.8 ± 57.4 (range 2,410-4,490) in the CD group.
DISCUSSION
The main feature of our study is that it included patients supplemented with calcium plus vitamin D as opposed to only vitamin D. The bone benefits deriving from this association have been confirmed in classical studies on osteoporosis, as shown in systematic reviews [11,12], although the issue is not without debate [13]. Also of interest in this study is the fact that we explored, for the first time, the impact of an intervention on specific developmental traits of the fetal femur at mid-gestation. This novel approach was possible thanks to advances in ultrasound technology, which allowed us to examine the specific structural response of the femur, an important element in the fetal skeleton.
We did not detect any effect of the association of vitamin D and calcium. This neutral effect is consistent with the lack of differences in birthweight between groups, a finding also reported in the literature in studies evaluating only vitamin D supplementation [14,4]. Even so, several questions arise.
One concerns the possibility that the intervention may have been too short in time and may have used doses that were too low. We supplemented with 1000 IU cholecalciferol per day, a dose in line with what is reported in recent studies [4]. That supplementation saw the baseline mean of 18.6 ng/mL increase to 24.9 ng/mL, and the percentage of women with levels above 20 ng/mL increase from 39% to 59%. However, 41% of the participants remained below the threshold of 20 ng/mL, which, applying the criteria used for the non-pregnant-state, corresponds to deficiency. This information is of interest because the circulating levels in the fetus are known to reflect those in the mother [15]. However, data from a combined cohort of two randomized supplementation trials in the US in which women received up to 4000 IU vitamin D3/day showed that the birthweight of neonates delivered by treated mothers did not differ from that seen in the usual care control group. Women treated with 4000 IU attained a mean 25OHD value of 41.9 ng/mL, which is almost twice that recorded in our cohort [14].
But that is not unanimous in the literature. The aforementioned study in India [3] detected a significant increase in birthweight. Of interest, though, the mean 25OHD in the mothers was only 23.5 ng/mL, similar to the value recorded our study. Similar positive results were found in Iran by investigators who attained 47.8 ng/mL in mothers assigned to the intervention group [16]. The possibility that socioeconomic level, lower in India and Iran as compared with the US, might have a conditioning effect is a possible explanation for the detected divergence. Against that interpretation, other investigators from the same countries failed to reproduce the claimed effect [17,18].
Another specific characteristic of our study was that usual care in our center meant supplementation with a poly-vitamin combination including 200 IU cholecalciferol. This intervention is unlikely to have obscured a potential effect of cholecalciferol as the circulating levels of 25OHD were significantly different between the two groups. Also, in our study, the intervention lasted 10 weeks, from the 20th to the 30th week of pregnancy. We focused on this period as it was the window we planned to examine with 3D ultrasound, as done in a previous study [8].The association of 1200 elemental calcium with 1000 IU cholecalciferol is an innovative aspect of our study. It ensured that the women in our treated group achieved the estimated average calcium intake recommended by scientific societies and governmental bodies during pregnancy [19,20]. Accordingly, it is very unlikely that any potential action of vitamin D was limited by insufficiencies in calcium intake. This is an important point, because while the impact of vitamin D supplementation on fetal growth is debated, a systematic review did detect some potential effect associated with calcium supplementation [6], something further suggested in a subsequent analysis [21]. It is also worth mentioning that our protocol associating calcium with cholecalciferol abrogated the risk of so-called maternal calcium metabolic stress, a specific status defined by a very low calcium intake, insufficient vitamin D, and elevated parathyroid hormone (PTH). The concatenated action of those three agents has been demonstrated to impact fetal growth [22]. It is of interest that babies born to mothers with calcium metabolic stress showed the features previously described in studies of vitamin D intervention in low socioeconomic environments, i.e., low birthweight and reductions in length and head circumference. Whether the concurrent supplementation of calcium might have limited that effect is an unproven assumption.In conclusion, in the context of an emerging interest in the impact of nutritional intervention on fetal growth, our study adds some data of interest. Our study is novel in two ways: first in that we added calcium to vitamin D, and second in that, thanks to modern ultrasound technology, we finely explored the intrauterine behavior of the fetal femur, an element offering crucial information for finely assessing fetal growth. Despite the low socioeconomic background of our group, which may be considered closer to that of women from less developed countries, we noted no discernible effect of the intervention. More studies evaluating protocols with varying dosages or treatment duration, as well as the investigation of other potential actors, like PTH, might help to clarify the issue.
CONFLICT OF INTEREST STATEMENT The authors declare that there is no conflict of interest.